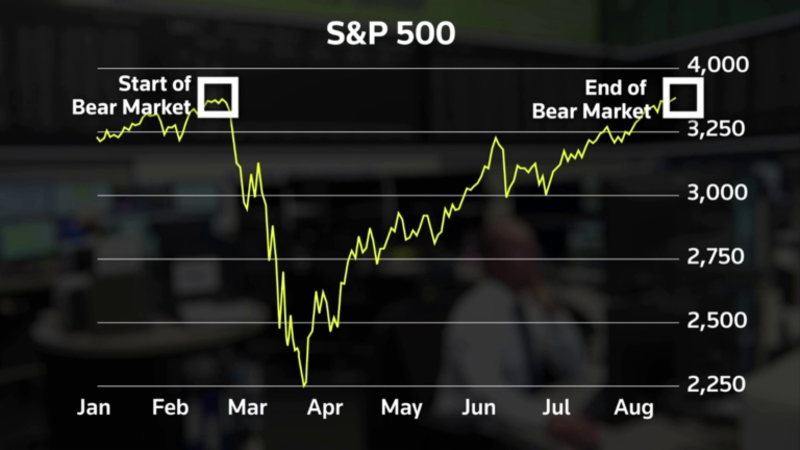
Think this was the article but they didnt allow link. posted here as fyi.
Actually this stuff looks like it could power CSL on.
CSL powers forward with plasma treatment research
For our free coronavirus pandemic coverage, learn more here.
By Emma Koehn
August 26, 2020 — 12.15am
Biotechnology giant CSL and its global colleagues are powering forward with the development of a coronavirus-fighting product that researchers hope could have advantages over the plasma transfusion treatment President Donald Trump announced approvals for this week.
The US Food and Drug Administration announced on Monday it had granted emergency use authorisation for convalescent plasma as a treatment for coronavirus. The treatment involves taking plasma, the yellow liquid in blood, from recovered coronavirus patients and transfusing this into those fighting COVID-19.
Biotech giants across the US are encouraging donors to come into centres and save lives in the face of the COVID-19 pandemic. CREDITENNY STEPHENS
The approval sparked controversy in research communities, with President Trump championing the strength of the treatment while a group of top US health officials including Dr Anthony Fauci said more evidence was needed to show the plasma worked.
There has been a global race to collect plasma from recovered coronavirus patients to study how it can be used to treat the disease. Earlier this year CSL joined a group of multinational biotechnology companies called the CoVIg-19 Plasma Alliance, to work on developing a plasma-based treatment for COVID-19.
CSL said that the FDA approval of plasma treatments in the US this week is unrelated to the product it is working on with its global colleagues. The treatment, called hyperimmune globulin, is a refined product that researchers hope could contain more virus-specific antibodies per unit than plasma for a straight transfusion.
Play video
1:03
FDA expands access to plasma treatment for COVID-19
President Donald Trump announced Sunday the emergency authorization of convalescent plasma for COVID-19 patients, in a move he called "a breakthrough." Other health experts said the treatment needs more study before it's celebrated. (Aug. 23)
"The clinical trial that we are participating in partnership with the NIH/NIAID [National Institutes of Health and National Institute of Allergy and Infectious Diseases] to evaluate our hyperimmune globulin (H-Ig) is unrelated to, and is unaffected by the FDA decision regarding emergency use authorisation for COVID-19 convalescent plasma for transfusion," CSL chief scientific officer Dr Andrew Nash said.
While plasma transfusions involve taking plasma from a recovered patient and transfusing this into those who are critically ill, H-Ig involves pooling the plasma of a number of recovered patients together. It is then processed to remove inactive viruses and concentrating the antibodies.
The alliance of companies has said there'sa hope the refined hyperimmune product will have a longer shelf-life and have more consistent antibody levels than plasma intended for straight transfusion.
"Because it is made from pooled convalescent plasma that has been purified and concentrated, H-Ig is standardised so it has a consistent level of antibodies in each unit," the CoVIg-19 Plasma Alliance said in an explanatory document.
RELATED ARTICLE
CORONAVIRUS PANDEMIC
Top US expert Anthony Fauci warns against premature release of vaccine
Add to shortlist
Trials of the study are still underway, however, and it's not known what the final data will show.
Dr Nash said the clinical trial that CSL was involved in was placebo controlled and randomised and it was hoped the study would show if the product was safe and effective against COVID-19.
"We have had discussions in advance with both the FDA and EMA [European Medicines Agency] to ensure the trial design meets their requirements. Additionally, as the H-Ig is a standardised and more concentrated amount of convalescent antibodies, we are hopeful this would translate into an even stronger efficacy signal," he said.
It's expected patients will be enrolled in a phase 3 trial of the treatment in the Northern Hemisphere this month.
Australian Red Cross Lifeblood is also collecting plasma from recovered donors locally for CSL to work on the first batch of product for a clinical trial of a similar plasma-based treatment in Australia at its Broadmeadows plant.
- Forums
- ASX - By Stock
- CSL
- Fda eua for convalescenct plasma
Fda eua for convalescenct plasma, page-8
-
-
- There are more pages in this discussion • 6 more messages in this thread...
You’re viewing a single post only. To view the entire thread just sign in or Join Now (FREE)
Featured News
Add CSL (ASX) to my watchlist
 (20min delay) (20min delay)
|
|||||
|
Last
$300.58 |
Change
-1.210(0.40%) |
Mkt cap ! $145.5B | |||
| Open | High | Low | Value | Volume |
| $300.74 | $300.99 | $298.81 | $240.1M | 820.5K |
Buyers (Bids)
| No. | Vol. | Price($) |
|---|---|---|
| 1 | 1000 | $300.56 |
Sellers (Offers)
| Price($) | Vol. | No. |
|---|---|---|
| $300.59 | 836 | 2 |
View Market Depth
| No. | Vol. | Price($) |
|---|---|---|
| 1 | 1000 | 300.560 |
| 1 | 15 | 299.920 |
| 1 | 1250 | 299.690 |
| 1 | 74 | 299.660 |
| 2 | 1250 | 299.540 |
| Price($) | Vol. | No. |
|---|---|---|
| 300.590 | 423 | 1 |
| 300.600 | 1250 | 1 |
| 300.630 | 204 | 1 |
| 300.660 | 426 | 1 |
| 300.740 | 486 | 1 |
| Last trade - 16.10pm 18/10/2024 (20 minute delay) ? |
Featured News
LGP
Record revenue of $10.2 million as European demand drives 40% quarterly growth, with $1 million in operating cash inflow and $0.5 million in cost savings forecast
| CSL (ASX) Chart |