I am well aware that the structure of this post may go a bit funny, so I have attached a PDF version including the data on FTO at this point. This is the second part of a 4 part series that will provide an update on Bisantrene and the chronicles of RAC. As we are all aware, the drug bisantrene has evolved over the last few years since the Rediscovery of Bisantrene review paper. My aim is to bring fresh understanding to old and more importantly new investors looking to get on board RAC. This work is 100% my own and from my own understanding. It is not financial advice.
Bisantrene Synthesis - DRAFT.pdf
3.0 Pillar 2 - Breast Cancer:
Breast cancer is the most commonly diagnosed cancer in the world, overtaking lung cancer last year, and is the leading cause of cancer deaths. It is estimated that there were 2.3 million global new cases of breast cancer in 2020. (https://pubmed.ncbi.nlm.nih.gov/33538338/). From those, there are an estimated 847, 952 new breast cancer cases that have arisen in Australia, Europe, and the US combined.
3.1 Treatment options for breast cancer
There are many treatment options for breast cancer and for the purpose of this review, I will focus on chemotherapy as it forms the basis of my rationale advocating and believing in the use of bisantrene as an alternative chemotherapeutic agent. One of the major concerns with chemotherapy treatment is the use of anthracyclines, like doxorubicin and epirubicin, and the resulting cardiotoxicity. So much so that anthracyclines have a maximum lifetime dose that doctors cannot exceed due to the increased risk of heart failure (Table 3). A study in the US found that the most common cause of death before or after 10 years was heart failure (Figure 2). What this means is the risk of future heart failure is causing doctors to shy away from the use of anthracyclines and chemotherapy for the treatment of breast cancer. In fact, due to the significant increase in heart failure risk, doctors will seldom use doses >250 mg/m2 (Figure 3). Figure 4 is a screenshot from an article exploring anthracycline use and cardiotoxicity and I thought the title of their section spoke volumes; “today’s cancer patients are tomorrow’s cardiac patients.” Within the article there is a link to another paper that discusses the decline in anthracycline use because of this phenomena - thus, could a safer anthracycline-based chemotherapeutic agent bring patients back?
Table 3: Maximal lifetime doses of anthracyclines

m2 indicates body surface area. https://www.ncbi.nlm.nih.gov/books/NBK538187/

Figure 2: A US study evaluating the cause of death in breast cancer patients.
(https://pubmed.ncbi.nlm.nih.gov/31840240/)

Figure 3: Long-term increase of heart failure risk with anthracycline dose
https://www.hindawi.com/journals/crp/2011/134679/

Figure 4: Today’s cancer patients are tomorrow’s cardiac patients
The demographics of those who receive a breast cancer diagnosis is part of the reason why I believe bisantrene will be so successful. Hear me out...the risk of a breast cancer diagnosis increases with age, with the average age of diagnosis falling around 63 years of age. This average age has increased from 57 years, indicating that those who are diagnosed with breast cancer are getting older and older. Among postmenopausal women (roughly say >55 years), those who are obese have a 20-40% increased risk of developing breast cancer when compared to normal-weight women. People aged 65 years and over are at the greatest risk of heart failure (in fact, it’s a leading cause of death among women), also postmenopausal obese women have 50% increased risk of heart failure than non-obese women.
So, what we have here is the convergence of multiple risk factors, which I will explain to you clearly. Current chemotherapeutic standard of care for breast cancer involves the use of anthracyclines, which are damaging for the heart. Those who are most at risk of developing breast cancer are those who are over the age of 63 and who are obese after experiencing menopause. However, women are at their greatest risk of heart failure after the age of 65, and postmenopausal obese women are 50% more likely to have heart failure than non-obese. What this means is the use of anthracyclines for breast cancer is increasing the risk of heart failure in a population that likely already has increased risk of heart failure, which, in my opinion, highlights the clinical significance of bisantrene. Calling on data discussed in the Rediscovery of Bisantrene paper (Figure 5).


Figure 5: Bisantrene safety profile
I would like you to pay special attention to the last sentence highlighted above and compare them to the maximal dose of other anthracyclines; 7080 mg/m2 of bisantrene with minimal cardiotoxicity compared to 550 mg/m2 of doxorubicin and likely lifelong increased risk of heart failure.
3.2 Bisantrene and breast cancer (2020 paper)
This paper tested the in vitro (performed outside of a living thing) efficacy of bisantrene alone, and in combination with cyclophosphamide (a standard drug used with anthracyclines in breast cancer therapy), in a range of human breast cancer cell lines that cover the major molecular and clinical subtypes, and span drug sensitive and resistant lines. The main results of the study are summarised below:
Bisantrene as a single agent is as cytotoxic as doxorubicin and epirubicin in a wide range of breast cancers
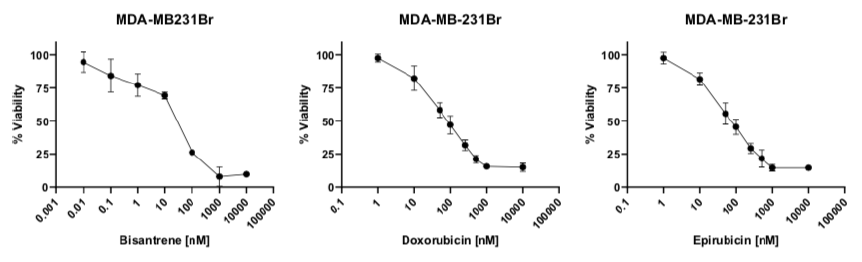
Bisantrene can kill breast cancer cell lines resistant to doxorubicin and epirubicin
Bisantrene acts similarly to doxorubicin and epirubicin when in combination with cyclophosphamide on breast cancer cells
The results of this study were good, but what they really did was confirm the historic data (Figure 3) collected for the use of bisantrene in breast cancer - highlighting the efficacy of bisantrene in comparison to cardiotoxic anthracyclines like doxorubicin and epirubicin. Notably, historic trials of bisantrene included heavily pretreated patients - basically, they were f*ed. Achieving a response in these groups was a significant effort and unfortunately, limitations of some of the trial designs led to unfavourable inferences made from the study.

Figure 3: Response rates in heavily pretreated breast cancer treatment failures
So, to put the previous section and this section all together into a neat little package. In a patient group with likely elevated risk of heart failure (obese + postmenopausal), bisantrene could become the safer and just as efficacious chemotherapeutic standard of care addressing up to 66% of the breast cancer market.
3.3 Breast cancer and FTO
Since 2015, there have been a number of associations made between FTO expression and breast cancer risk. As it stands, the current evidence suggests that FTO is overexpressed in breast cancer cells which leads to a range of biological functions including cancer cell survival and colony formation, cell energy metabolism, proliferation, and metastasis.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7751723/pdf/ott-13-12845.pdf
A more recent systematic review (the highest quality research that can be done) has investigated the role of estrogen in the association between FTO gene polymorphisms and breast cancer in 16 different journal articles. The main conclusion confirming the association between breast cancer and FTO. The study raised an additional point regarding estrogen and FTO expression, suggesting that estrogen levels of obese postmenopausal women were higher and led to increased FTO expression.
This information is a catalyst for thought. Typically FTO inhibition would use low-dose bisantrene, however, the majority of trials have used much larger doses of bisantrene (in the mg/m2 range). Would an effective treatment of breast cancer come from a combination of low and high dose bisantrene? It is definitely food for thought.
3.4 Total addressable market
There are multiple different types of breast cancer, including triple negative, HER2+, and luminal A and B. Figure 4 below shows the four different breast cancer types, as well as the use of chemotherapy for each type. Up to 66% of breast cancer patients will receive chemotherapy, with the majority of those diagnosed with stage 3 or 4 breast cancer (Table 1). Table 2 provides a summary of data collected on the rates of different stages among women 40-74 years. This information indicates that approximately 75-80% of breast cancer patients are in stages 2-4 and would be a potential target for chemotherapy. Together, this information tells us that chemotherapy is used in a wide range of cancers (triple negative, HER2+, and Luminal B) typically towards the later stages of development.
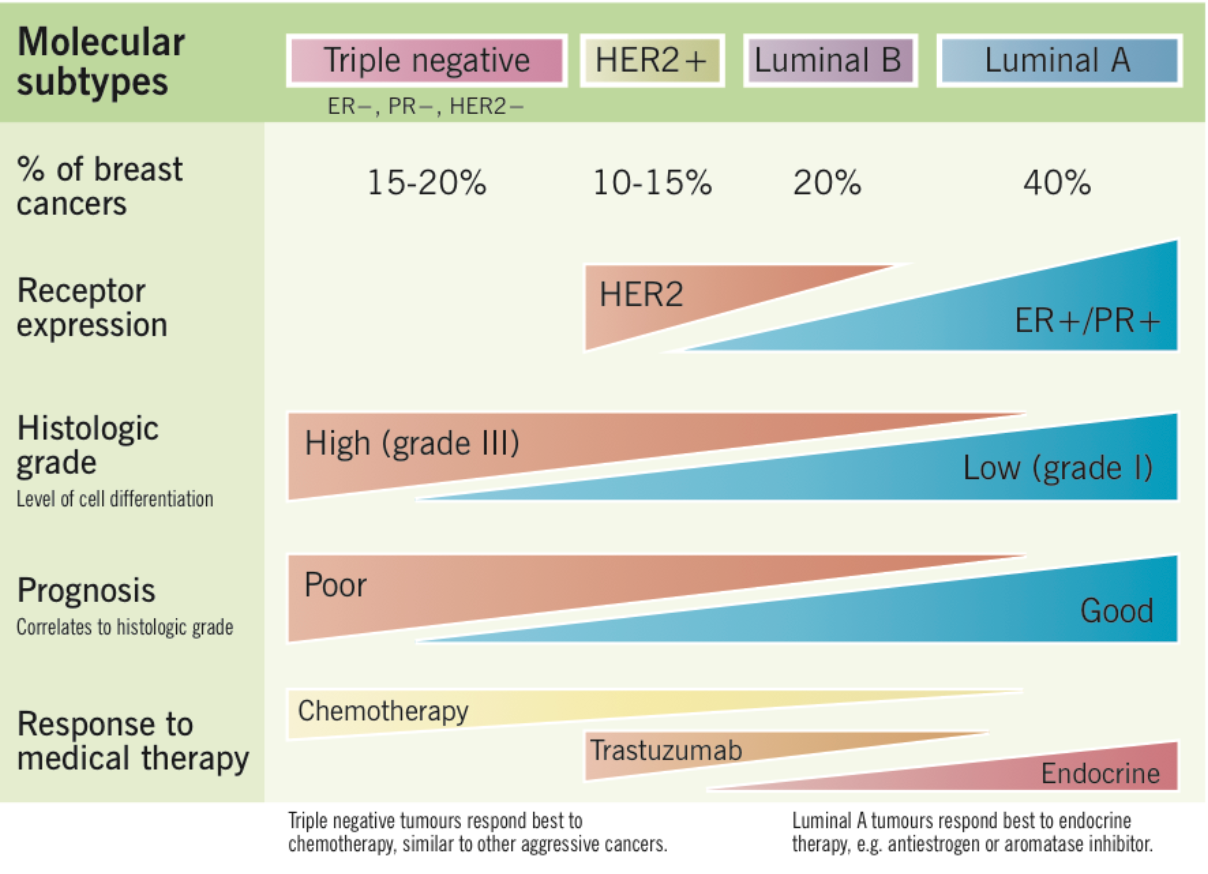
(https://www.grepmed.com/images/3513...agnosis-molecular-incidence-subtypes-invasive)
Figure 4: Breast cancer types and medical therapy use.
Table 1: Chemotherapy treatment rates for breast cancer patients
Cancer Type
Stage 1
Stage 2
Stage 3
Stage 4
Breast
0
17%
62%
66%
(https://www.asbestos.com/treatment/chemotherapy/statistics/)
Table 2: Prevalence of breast cancer by stage
Cancer Type
Stage 1
Stage 2
Stage 3
Stage 4
Breast
21-25%
44-53%
21-27%
4-5%
(https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6312406/)
It is at this point where I flex my weakest muscles and try on some valuing. I’m going to do the best Wombat I can. Hopefully, doing some rough calculations will provide some detail about the potential, addressable market for breast cancer. So I don’t lose anyone, I'm going to lay out all of the steps I went through to come to my share price estimates at Figure 5.
These are the assumptions I have made:
Use of 60% molecular subtype appropriate.
Lower end of breast cancer stage incidence for stage 2, 3, and 4 (44%, 21%, and 4% respectively) to be conservative.
Used chemo treatment rates identified above for breast cancer stage 2, 3, and 4 (17%, 62%, and 66%).
The average price per year ($56,902) for cancer drugs was estimated by using averages taken from a global comparison of cancer drugs and then weighting them to the different regions (https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5641070/)
Percentage market share for bisantrene set to 15%, which is based on previous analysis of bisantrene as a safer alternative to anthracycline. I believe that the safety profile of bisantrene will attract new patients and thus have also included a more reasonable (in my opinion) 30% share and 45% share of the discounted breast cancer population.
Buyout Multiple determined by the following sections buyout multiple of 4.5 for Immunomedics
Fully diluted shares on issue to 200M to be conservative.
At 15%, 30%, and 45% of the addressable market for bisantrene, my share price estimates are $27.8, $55.5, and $83.3 respectively. Note: these figures are highly speculative.

Figure 5: Total value of addressable market for breast cancer.
3.5 Breast cancer drug buyout - a comparison of market depth and value
I would like to bring to the reader's attention Immunomedics. They were bought out by Gilead in September of 2020 for USD $21B for their main drug Trodelvy. The catalyst for this purchase was their phase 3 ASCENT study being halted by the Independent Data Safety Monitoring Committee (IDSMC), as their drug significantly improved progression-free survival (PFS) and overall survival (OS) in previously treated patients with advanced mTNBC. The IDSMC halting your trial early is indicative of fantastic results and is something any aspiring biotech company would want from their drug. Why is this important? Let’s see:
Analysts have suggested that Troveldy could generate USD $5B per year in peak revenue sales, indicating that Immunomedics were acquired for roughly 4.2x their estimated peak revenue. The type of breast cancer that their drug targets is specialised - metastatic triple negative breast cancer, which accounts for 15-20% of the addressable breast cancer market. That means based on the data I presented in the previous section and before we have even got to breast cancer stages, Trodelvy has the potential to treat approximately 170,000 people at best. Now, of those 170,000 people, they have to be metastatic, which is likely stage 4 breast cancer, and as part of their FDA approval, patients have to have failed two prior treatments. It’s at this point that I would like to remind you that chemotherapy (bisantrene) was applicable in roughly 60% of the breast cancer market.
Because I know how many of you love seeing a figure, I will make a very simple comparison of bisantrene to immunomedics based on peak revenue potential and percentage of addressable market (Figure 6).
Assumptions:
Assumed RAC market and revenue estimates are x3 immunomedics from previous sections data
Assumed immunomedics buyout multiplier for RAC
Clinical probability success very low at 20% to be conservative
Fully diluted shares on issue at 200M allowing for extra dilution

Figure 6: Share price estimate from immunomedics buyout and peak sales
Based on the peak sales and target market of immunomedics, I value the potential of bisantrene at $79.3 per share.
3.6 Breast Cancer Conclusion
You need to make your own mind about the use of bisantrene as a chemotherapeutic agent for breast cancer. Although, I firmly believe that with the opportunity to address 60% of the breast cancer market, with a drug that will be as efficacious and safer than the current anthracyclines in a patient group with likely preexisting increased risk of heart failure, bisantrene is set to make its mark on breast cancer therapy in westernised worlds. The relationship between FTO and breast cancer is all the more enticing, as if bisantrene is proven in the clinic for melanoma during year 2021, there may be enough of an indication to run a trial examining high and low dose bisantrene for breast and/or other cancers. The dualistic dose approach to breast cancer treatment would be, based on my best knowledge, novel and revolutionary.
 (20min delay)
(20min delay)




