MARCH 1, 2024
11 MIN READ
These Cancers Were Beyond Treatment—But Might Not Be Anymore
New drugs called antibody-drug conjugates help patients with cancers that used to be beyond treatment
BY JYOTI MADHUSOODANAN
Keith Negley
March 2024 Issue
Pharmaceuticals
In the long and often dispiriting quest to cure cancer, the 1998 approval of the drug Herceptin was a tremendously hopeful moment. This drug for breast cancer was the first to use a tumor-specific protein as a homing beacon to find and kill cancer cells. And it worked. Herceptin has benefited nearly three million people since that time, dramatically increasing the 10-year survival rate—and the cancer-free rate—for what was once one of the worst medical diagnoses. “Honestly, it was sort of earth-shattering,” says oncologist Sara M. Tolaney of the Dana-Farber Cancer Institute in Boston.
But the drug has a major limitation. Herceptin's beacon is a protein called HER2, and it works best for people whose tumors are spurred to grow by the HER2 signal—yet that's only about one fifth of breast cancer patients. For the other 80 percent of the approximately 250,000 people diagnosed with the disease every year in the U.S., Herceptin offers no benefits.
The hunt for better treatments led researchers to reimagine targeted therapies. By 2022 they had developed one that linked Herceptin to another cancer-killing drug. This therapy, for the first time, could damage tumors that had vanishingly low levels of HER2. The drug, named Enhertu, extended the lives of people with breast cancer by several months, sometimes longer. And it did so with fewer severe side effects than standard chemotherapies. The U.S. Food and Drug Administration approved its use in that year.
The news got even better in 2023. Researchers reported that Enhertu appeared to work even on tumors with seemingly no HER2 at all. (It's possible the cancers did have the protein but at very low levels that escaped standard detection methods.) “Exciting!” says oncologist Shanu Modi of Memorial Sloan Kettering (MSK) Cancer Center in New York City, who helped to run the study that led to Enhertu's approval. “They did this provocative test and saw this almost 30 percent response rate” in tumors apparently lacking the cancer protein, she notes.
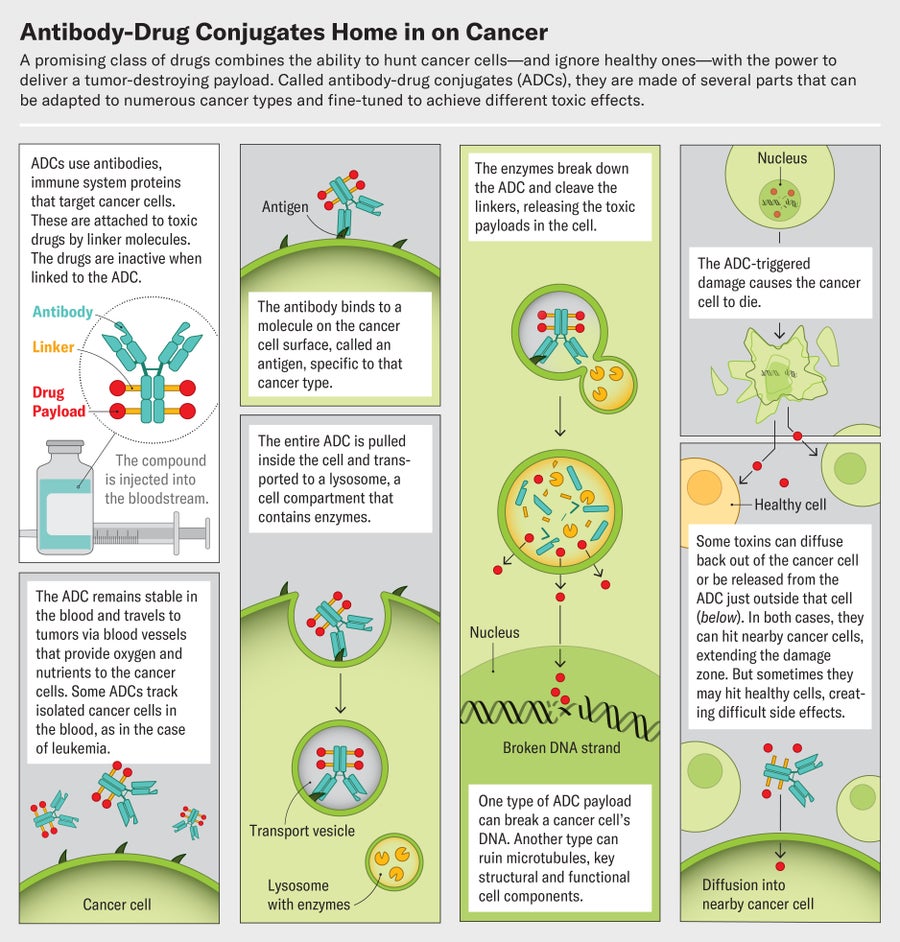
Enhertu belongs to an ingenious and growing class of targeted cancer drugs called antibody-drug conjugates, or ADCs. The compounds are built around a particular antibody, an immune system protein that homes in on molecules that are abundant on cancer cells. The antibody is linked to a toxic payload, a drug that kills those cells. An ADC's affinity for cancer means it spares healthy cells, avoiding many of the side effects of traditional chemotherapy. And each antibody can be paired with several different drugs. This Lego-like assembly opens up a world of mix-and-match possibilities. Researchers can use the same drug to treat many cancers by switching up the antibody, or they can attack one type of tumor with many different ADCs that target several cancer biomarkers on the cells. This ability “changes the way we think about drug development,” Tolaney says.
The idea for ADCs is not entirely new—the first one was cleared for patient use in 2000—but recently scientists have learned intricate chemical construction techniques that make the compounds much more effective, and they have identified new cancer-specific targets. These advances have driven a wave of new development. Fourteen ADCs have been approved for breast, bladder, ovarian, blood, and other cancers. Approximately 100 others are in the preclinical pipeline. One ADC for breast cancer, known as T-DM1, proved much more effective than Herceptin and has now become the standard of care for early stages of disease. “It is pretty cool to see how things have changed so quickly,” Tolaney says. Buoyed by the successes, researchers and pharmaceutical companies are pouring resources into developing more powerful ADCs—perhaps even ones that can work across a wide range of cancer types. Pharma giants such as Gilead, Roche and BioNTech have invested heavily in their ADC programs; in October 2023, for example, Merck put $4 billion into a partnership with Daiichi Sankyo, the biotechnology firm that partnered with AstraZeneca to produce Enhertu.
Does Long-Term Benadryl Use Increase Dementia Risk?
HANNAH SEO
But the new drugs are still beset by some mysterious problems. Some ADCs have side effects similar to those caused by traditional chemotherapies—which shouldn't happen, because the drugs are supposed to target cancer cells alone. On patient forums, people describe needing to reduce their doses because of intolerable nausea or fatigue. These drawbacks limit ADCs' use, so scientists and pharma companies are urgently trying to figure out what is causing them.
In the clinical trial that led to Enhertu's approval, patients typically had already received different kinds of chemotherapy drugs, such as medications that stop cells from multiplying. But these drugs—and other forms of chemotherapy—do not distinguish between a cancer cell and a healthy one. Any cell trying to make DNA or multiply is vulnerable, and normal tissue as well as tumors can be attacked. Fully 64 percent of people on standard chemotherapy experience nausea, diarrhea, fatigue, and other negative side effects. For many, these can be as debilitating as cancer itself. Such effects limit the dose people can take and the length of treatment, leaving windows of opportunity for tumors to grow resistant and rebound.
For many years researchers have sought less toxic alternatives, envisioning precision drugs that target cancers and spare healthy cells. The idea of ADCs sprang from the exquisite specificity of antibodies. If highly toxic forms of chemotherapy could be strapped onto antibodies, the toxins would reach only the cancer cells and no others. Although the concept was straightforward, attempts at making ADCs faltered for decades.
Some of the earliest attempts used drugs that just weren't strong enough. In the 1950s, for instance, researchers linked a drug named methotrexate to an antibody that targets carcinoembryonic antigen, a common tumor marker, and tested whether the construct could treat advanced colorectal and ovarian cancers in people. The drug bound to its target but had little therapeutic effect. Researchers then swung too far to the other end of the spectrum and tried using much more toxic drugs instead. But these drugs triggered serious side effects.
Credit: Jen Christiansen; Graphics consultant: Greg Thurber/University of Michigan
Greg Thurber, a chemical engineer at the University of Michigan, looked into this conundrum. He began working on ADCs when studying how antibodies spread through the body to bind to their targets. After ADCs infiltrate a tumor through its network of blood vessels, the compounds slip out of these vessels and into cancer cells to kill them, Thurber says. But the ADCs that existed at the time never got past the cells just outside the blood vessels. They bound too tightly. The key to improved effects, it turned out, was tailoring the antibody parts so they zeroed in on cancer cells but had a loose enough grip for some to slip into the interior of the tumor. “A lot of people in the field had a very simple concept—we put a chemotherapy drug on an antibody, it targets it to the cancer cell, and it will avoid healthy tissue,” Thurber says. “That's not at all how they work in reality.”
Tinkering with the drug component of ADCs, as well as the antibody, eventually led to a cancer-killing sweet spot. In 2013 the fda greenlit T-DM1 for breast cancer. Its antibody is trastuzumab (the “T” in T-DM1), the same antibody used in Herceptin. The drug attached to this antibody is notable because it's too dangerous to be used on its own. Known as emtansine, it was initially discovered in the 1970s but shelved because it was too toxic to too many cells. Tethered together as T-DM1, however, the drug and antibody generally stayed away from healthy cells and proved to be a potent and precise combination.
Sign Up for Our Daily Newsletter
Email Address
By giving us your email, you are agreeing to receive the Today In Science newsletter and to our Terms of Use and Privacy Policy.
Sign Up
Thank you for signing up!
Check out our other newsletters
In the early 2000s Modi helped to conduct a trial of T-DM1—branded Kadcyla by its maker, Genentech—in people who had an especially difficult disease: advanced HER2-positive breast cancer that had spread throughout the body. Only those who had run out of other treatment options were enrolled. “We were taking people who in some cases were really looking to go to hospice,” Modi says. Yet “almost every patient who was enrolled on that drug had benefits. It was really so satisfying.”
In another trial of about 1,500 people with early breast cancer, an interim data analysis, published in 2019, estimated that 88 percent of those who received T-DM1 would be cancer-free three years later, compared with just 77 percent of those who received Herceptin alone. The drug has proved “more active than most of the therapies we were giving to patients, and it was associated with a better safety profile,” Modi says.
Kadcyla's success against difficult-to-treat cancers didn't just transform some patients' lives. It pumped enthusiasm—and, perhaps more important, pharmaceutical industry dollars—into the idea of ADCs. Researchers now knew that when pieced together correctly, it was possible to load an antibody with drugs too toxic to be used otherwise and still produce a medicine that worked better than traditional chemotherapy.
Several similarly designed ADCs have been approved for a range of different cancer types. Many of these carry drugs that inhibit the enzyme topoisomerase 1, which is essential for DNA replication. Like emtansine, the drug used in Kadcyla, newer topoisomerase inhibitors are too toxic to be used as freestanding drugs but are much less harmful when they're largely restricted to tumor cells. And Kadcyla itself, after being shown to slow or stall late-stage breast cancer, is being tested on patients with very early-stage disease to see whether treatment at that point can not only slow cancer down but actually cure it. Its success “was sort of the catalyst for continued exploration,” Modi says. “Can we build on this? Can we do even better?”
Doing better, it turns out, involves designing good linker molecules that tie the antibody to the drug. These tiny structures act like chemical triggers. They must remain perfectly stable until they reach their target, then unclip from the antibody to discharge their payload at the tumor. Some of the earliest attempts at making ADCs failed not because of their antibodies or drugs but as a result of unstable linkers.
Modern ADCs rely on two types of linkers. One kind remains unbroken even when the ADC reaches its target. The other kind, known as cleavable linkers, are chemicals that break in response to very specific cues, such as enzymes that are abundant in tumors, in the spaces between individual cancer cells. Once an ADC is within the tumor's boundaries, these enzymes cleave the linker and release the drug payload.
Cleavable linkers are showing impressive advantages, and more than 80 percent of currently approved ADCs now use them. An ADC with a noncleavable linker will kill only the cell it attaches to, but one that splits up could place drug molecules near neighboring tumor cells and destroy them as well. This so-called bystander effect can make the drugs much more effective, Thurber says.
Enhertu, for instance, uses the same antibody as Kadcyla but with a cleavable linker (Kadcyla uses a noncleavable version) and a different drug. Each Enhertu antibody carries approximately eight drug molecules, compared with about three per antibody in Kadcyla. In one recent study, researchers compared the effects of these two drugs in people with HER2-positive breast cancers. Enhertu was the clear winner. It stopped tumor growth for more than two years on average, whereas Kadcyla did so for just six months. “It was a landslide in terms of how much better it was,” Tolaney says. “It's a really nice example of how ADC technology leads to dramatic differences in outcomes.”
The bystander effect also explains, in part, why Enhertu is effective against tumors that have barely any HER2: once the ADC enters a tumor and the drug molecules detach, they can kill neighboring tumor cells even if those bystanders don't carry much HER2 on their surface. This action, along with the use of a diagnostic test that can miss extremely low HER2 levels, could explain the results from the trial where the drug seemed to work on tumors with no HER2. That trial employed an assay known as an IHC test. It is generally used to categorize cancers as HER2 positive or negative, not to measure the amount of the protein present. A negative result typically means 10 percent or fewer of the tumor's cells have HER2 on their surfaces. Yet 10 percent may be enough to attract a few Enhertu particles, and the bystander effect might be sufficient to destroy tumor cells, Modi says.
Enhertu is not the only ADC that appears to work this way. In a 2022 study, researchers found that Trodelvy, an ADC that targets a surface protein known as TROP2, seemed to be more effective than standard chemotherapy for people with metastatic triple-negative breast cancer, a particularly hard-to-treat disease. Trodelvy was better irrespective of how much or how little TROP2 was detected on tumors. “That, to me, is wild,” Tolaney says. “We're excited about it because these cancers are having benefits [apparently] without the target.”
This new generation of ADCs is making a difference in other types of cancers previously thought to be intractable, such as metastatic bladder cancer. In 2021 the fda approved Trodelvy and another ADC named Padcev to treat this illness. For 30 years the standard of care for this type of bladder cancer was chemotherapy alone, says oncologist David J. Benjamin, who treats genitourinary cancers at Hoag Family Cancer Institute in southern California. “Now we have multiple new treatments, and two of them happen to be antibody-drug conjugates,” Benjamin says. In clinical trials for patients with advanced bladder cancer, Padcev combined with a drug that stimulates the immune system shrank tumors or stalled their growth in more than 60 percent of people. In a whopping 30 percent of those who received the two-drug combination, their cancer completely disappeared—an unprecedented success.
But even newer ADCs aren't without problems. The bystander effect, which makes them so effective, can spread far enough from the tumor to affect healthy cells, causing hair loss, nausea, diarrhea, fatigue, and other side effects that are disturbingly similar to the fallout of old-school chemo. ADCs also have been linked to a variety of eye problems ranging from conjunctivitis to severe vision loss.
Another explanation for these nasty effects is that there are no protein targets that are exclusive to cancer cells. These proteins, also known as antigens, are more abundant in cancers but may appear in normal cells. That makes some binding of ADCs to healthy cells unavoidable. “I can't think of any examples of true tumor-specific antigens,” says Matthew Vander Heiden, a molecular biologist at the Koch Institute at the Massachusetts Institute of Techonology. Further, ADCs, like any other medicine or antibody, are eventually ingested and metabolized by noncancerous cells. This process fragments them into smaller pieces, releasing payload drugs from their linkers and triggering reactions.
Still, the ability to take ADCs apart and tweak their components—something that isn't possible with traditional treatments—offers researchers the chance to find versions with fewer side effects and more advantages. At present, most ADCs are used at the maximum dose a person can tolerate. That might not be true with future versions. When developing a medication, whether it's a simple painkiller, a chemotherapy or an ADC, researchers begin by figuring out the lowest dose at which the drug is effective. Then they work out the highest dose that people can receive safely. The space between those two doses, known as a therapeutic window, is usually small. But the ability to swap components offers ADC researchers many routes to widening it. Eventually drugmakers might create ADCs so effective that patients never need to take the highest tolerable dose—a much lower one would eliminate tumors without creating unintended consequences such as nausea or hair loss.
Shifting away from toxic chemotherapy-based drugs as payloads could also reduce side effects. Some recently approved ADCs, for instance, link antibodies to drugs that can activate the body's own immune system to attack cancer cells rather than relying on cell-poisoning chemicals. In addition, scientists are exploring ways to deliver radiation therapy directly to tumors by tethering antibodies to radioisotopes. Joshua Z. Drago, an oncologist at MSK Cancer Center, says that with the right kind of linkers, ADCs “could theoretically deliver any kind of small-molecule medication.”
Ultimately, recombined and improved components could lead to the type of swap that cancer patients really care about: exchanging their disease for a cure.
FROM OUR ARCHIVES
Antibody-Drug Conjugates and Cancer Treatment: Making “Smart Bombs” Smarter. Trevor Hallam; ScientificAmerican.com, July 4, 2013.
Scientific American.com/archive
JYOTI MADHUSOODANAN is a health and science journalist based in Portland, Ore. She has a Ph.D. in microbiology.
More by Jyoti Madhusoodanan
This article was originally published with the title “Targeting Cancer, Sparing Patients” in Scientific American Magazine Vol. 330 No. 3 (March 2024), p. 36
doi:10.1038/scientificamerican0324-36
View This Issue
Popular Stories
PUBLIC HEALTH APRIL 2, 2024
Are Your Solar Eclipse Glasses Fake? Here’s How to Check
You’re going to want to guard your eyes from the sun using legitimate protective gear
SARAH SLOAT
ASTRONOMY FEBRUARY 2, 2024
Here Are the Best Places to View the 2024 Total Solar Eclipse
Weather predictions and population statistics show the best spots to see the total solar eclipse over North America this April
KATIE PEEK
WEATHER APRIL 3, 2024
The Latest Weather Forecast along the Total Solar Eclipse Path
From cloud coverage to clear skies, here’s up-to-date weather conditions expected along the path of April 8’s total solar eclipse
ANDREA THOMPSON
CONSERVATION APRIL 4, 2024
Decades-old Cans of Salmon Reveal Changes in Ocean Health
Researchers used tinned fish to reconstruct parasitic population change, giving new meaning to the phrase “opening a can of worms”
RACHEL NUWER
CLIMATE CHANGE APRIL 4, 2024
Geoengineering Test Quietly Launches Salt Crystals into Atmosphere
A solar geoengineering experiment in San Francisco could lead to brighter clouds that reflect sunlight. The risks are numerous
CORBIN HIAR, E&E NEWS
ANIMALS MARCH 11, 2024
A Double Emergence of Periodical Cicadas Isn’t Cicada-geddon—It’s a Marvel
The U.S. will see two adjacent broods of periodical cicadas emerge this spring
MEGHAN BARTELS
Expand Your World with Science
Learn and share the most exciting discoveries, innovations and ideas shaping our world today.
SubscribeSign up for our newslettersSee the latest storiesRead the latest issue
Follow Us:
Scientific American is part of Springer Nature, which owns or has commercial relations with thousands of scientific publications (many of them can be found at www.springernature.com/us). Scientific American maintains a strict policy of editorial independence in reporting developments in science to our readers.
© 2024 SCIENTIFIC AMERICAN, A DIVISION OF SPRINGER NATURE AMERICA, INC.
ALL RIGHTS RESERVED.
Apr 26, 2023
HER2-targeted DEP® SN-38 ADC outperforms in HER2+ human cancer model (ASX Announcement)
Melbourne, Australia; 26 April 2023: Starpharma (ASX: SPL, OTCQX: SPHRY) today announces that it has developed a HER2-targeted DEP® SN-38 antibody-drug conjugate (ADC), which has shown significant anti-tumour activity in a HER2+ human ovarian cancer xenograft model, outperforming the approved ADC product, Enhertu®[2].
- HER2[1] is an important target in a number of different cancers, with antibody-drug conjugates (ADCs) targeting this receptor, including AstraZeneca’s marketed Enhertu®, showing significant promise
- Starpharma has developed a HER2-targeted DEP® ADC, utilising the active metabolite of irinotecan, SN-38
- Starpharma’s HER2-targeted DEP® SN-38 ADC outperformed Enhertu®, showing significantly greater anti-tumour activity and improved survival in a HER2+ human cancer xenograft model
ADCs represent an innovative and growing area of cancer treatment, with encouraging clinical advances and product approvals in recent years. The global ADC market grew from USD ~$5.8 billion in 2021 to USD ~$8.0 billion in 2022 and is projected to reach USD ~$22.9 billion in 2030[3]. HER2 is an important target for cancer treatments, with ADCs targeting this receptor showing significant promise. Currently marketed HER2-targeted ADCs include Kadcyla® (Genentech and Roche) and Enhertu® (AstraZeneca and Daiichi-Sankyo), which is now approved for use in patients with HER2+ or HER2-low metastatic breast cancer, HER2+ metastatic gastric or gastroesophageal junction cancer, and HER2-mutant metastatic non-small-cell lung cancer[4].
Starpharma’s HER2-targeted DEP® SN-38 ADC comprises a HER2-directed antibody, trastuzumab, linked to DEP® dendrimers loaded with the topoisomerase I inhibitor, SN-38, which is the active metabolite of irinotecan. Starpharma’s HER2-targeted DEP® SN-38 ADC has been designed with a higher drug-to-antibody ratio (DAR), or drug loading, than currently marketed ADCs.
The anti-cancer activity of Starpharma’s HER2-targeted DEP® SN-38 ADC was demonstrated in an established HER2+ human cancer xenograft model, utilising the SKOV-3 ovarian cancer cell line that overexpresses HER2. The HER2-targeted DEP® SN-38 ADC and Enhertu®, dosed intravenously (IV) on study days 1, 8 and 15, significantly inhibited tumour growth compared with saline control. The anti-tumour effect of the HER2-targeted DEP® SN-38 ADC was statistically significantly greater than the anti-tumour effect of Enhertu® over the duration of the study (p<0.0001). All animals in the HER2-targeted DEP® SN-38 ADC group survived to the end-of-study and survival was statistically significantly greater for the HER2-targeted DEP® SN-38 ADC-treated animals compared with Enhertu® and the saline control group (p<0.0002).
Starpharma’s HER2-targeted DEP® SN-38 ADC, containing SN-38, achieved superior tumour growth inhibition and survival compared with Enhertu®, despite the active drug in Enhertu®, DXd, being approximately 10 times more potent against topoisomerase I than SN-38[5].
Key advantages of Starpharma’s DEP® platform for ADCs include its ability to achieve higher DAR, and therefore higher drug payload, than conventional ADCs (see Table 1, below); its greater flexibility in terms of linker strategies to precisely control drug release profiles; and its ability to widen the therapeutic index of toxic drug payloads. In addition, Starpharma’s DEP® platform provides unique flexibility in terms of compatible targeting agents, including biologics (whole antibodies and fragments), small molecules, peptides and other approaches.
Table 1. HER2 ADCs Drug-to-Antibody Ratios (DAR), Drug Payload and Payload Mechanism of Action
HER2 ADC
Approximate Drug-to-Antibody Ratio (DAR)
Drug Payload
Payload Mechanism of Action
Kadcyla®
(Genentech/Roche)
3.5
DM-1 (emtansine)
Microtubule
inhibitor
Enhertu®
(AstraZeneca/Daiichi-Sankyo)
8
DXd (exatecan derivative)
Topoisomerase I inhibitor
HER2-targeted DEP®
SN-38 ADC
(Starpharma)
13
SN-38
Topoisomerase I inhibitor
Starpharma CEO, Dr Jackie Fairley, said: “Antibody-drug conjugates are one of the fastest growing classes of anti-cancer drugs, with significant progress being made in recent years, including the recent approval and clinical success of Enhertu®, which has shown significant promise in helping patients with HER2+ cancers.
“Starpharma is pleased to report this new data for its internally developed HER2-targeted DEP® SN-38 ADC demonstrating significant anti-tumour activity and survival, compared to Enhertu®, in a HER2+ cancer model.
“Starpharma’s DEP® technology delivers a number of advantages in the design of innovative ADCs, including the ability to load more drug payload molecules per construct and having greater flexibility in linker strategies. In addition to our internal activities in this area, Starpharma is delighted to be working in partnership with a number of companies, including MSD, to develop dendrimer-based ADCs using Starpharma’s DEP® technology.”
Study Methods
Starpharma evaluated the anti-cancer activity of HER2-targeted DEP® SN-38 ADC compared to Enhertu® in an established HER2+ human cancer xenograft model. The study was conducted at the Monash Institute of Pharmaceutical Sciences (MIPS).
This murine xenograft study used the HER2+ SKOV-3 ovarian cancer cell line, in NOD-SCID-Gamma (NSG) immunodeficient mice. The SKOV-3 cell line was chosen because it naturally overexpresses HER2 and is not dependent on estrogen for growth and survival, meaning that it is a robust model for evaluation of HER2-targeting drugs. Tumour cells were inoculated subcutaneously and the resultant tumours were measured 2-3 times weekly using electronic calipers. Tumour volume (mm3) was calculated at each timepoint.
Following tumour establishment, groups of mice (n = 7-8 per group) were dosed IV on days 1, 8, and 15 as follows:
Study Results
- Vehicle (saline)
- Control dendrimer-SN-38[6] – 3.28 mg/kg (SN-38 equivalent)
- Enhertu® – 5 mg/kg (total construct dose)[7]
- HER2-targeted DEP® SN-38 ADC – 3.28 mg/kg (SN-38 equivalent)
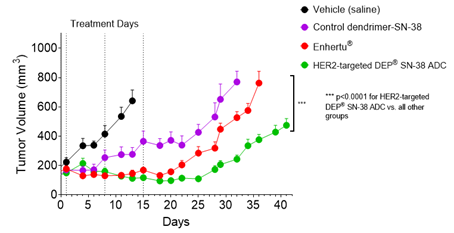
Effect of HER2-targeted DEP® SN-38 ADC vs. Enhertu® on Tumour Volume Over Time
Figure 1. HER2-targeted DEP® SN-38 ADC achieved statistically significantly enhanced tumour growth inhibition compared with Enhertu® or the control dendrimer that lacked the HER2-targeting antibody (p<0.0001 for both comparisons). Points and error bars show means ± standard error of the mean (SEM). Statistical analyses used the Mixed Effects Model (GraphPad Prism v9.4.1).
Kaplan-Meier Survival Curve
Figure 2. Analysis of survival curves showed significantly enhanced probability of survival of the HER2-targeted DEP® SN-38 ADC group versus all other groups (p<0.0002). Probability of survival in the HER2-targeted DEP® SN-38 ADC group was 100% (at end-of-study, 45 days). Median survival, or the time at which probability of survival dropped to 50%, was 40 days for Enhertu®, 28 days for the control dendrimer, and 13 days for the vehicle control. Survival analyses used tumour volume (≥ 1000 mm3), tumour-site ulceration, or end-of-study as endpoints.
Survival analyses used the logrank (Mantel-Cox) test (GraphPad Prism v9.4.1).
All treatments were well tolerated, as measured by change in body weight over time.
View the ASX Announcement here.
[1] Human epidermal growth factor receptor 2
[2] ENHERTU® is a registered trademark of Daiichi Sankyo Company Limited. Enhertu® comprises the humanised monoclonal antibody/HER2-directed antibody, trastuzumab, covalently linked to a topoisomerase I inhibitor payload, DXd (an exatecan derivative)
[3] https://www.grandviewresearch.com/industry-analysis/antibody-drug-conjugates-market
[4] https://www.enhertu.com
[5] Ogitani Y, Aida T, Hagihara K, et al. DS-8201a, A Novel HER2-Targeting ADC with a Novel DNA Topoisomerase I Inhibitor, Demonstrates a Promising Antitumor Efficacy with Differentiation from T-DM1. Clin Cancer Res. 2016;22(20):5097-5108. doi:10.1158/1078-0432.CCR-15-2822
[6] Control dendrimer-SN-38 = DEP® dendrimer with SN-38 covalently attached (the same dendrimer and linker used to make DEP® SN-38 ADC but lacking the HER2 targeting moiety, i.e., trastuzumab).
[7] In preclinical studies, Enhertu® has been used at doses ranging from 3-10 mg/kg IV. The area under the curve (AUC) in mice at 5 mg/kg Enhertu® is estimated to be comparable with that of Enhertu® in humans at the clinical dosage, and hence 5 mg/kg was selected as the dose for the current study.
This contains certain forward-looking statements.
About Starpharma
© Starpharma Holdings Limited 2021
- Forums
- ASX - By Stock
- HER2-targeted DEP® SN-38 ADC outperforms in HER2+ human cancer model
SPL
starpharma holdings limited
Add to My Watchlist
4.00%
 !
12.0¢
!
12.0¢
MARCH 1, 2024 11 MIN READ These Cancers Were Beyond...
Featured News
Add to My Watchlist
What is My Watchlist?
A personalised tool to help users track selected stocks. Delivering real-time notifications on price updates, announcements, and performance stats on each to help make informed investment decisions.
 (20min delay) (20min delay)
|
|||||
|
Last
12.0¢ |
Change
-0.005(4.00%) |
Mkt cap ! $50.18M | |||
| Open | High | Low | Value | Volume |
| 12.0¢ | 12.5¢ | 11.5¢ | $3.755K | 31.66K |
Buyers (Bids)
| No. | Vol. | Price($) |
|---|---|---|
| 2 | 63402 | 11.5¢ |
Sellers (Offers)
| Price($) | Vol. | No. |
|---|---|---|
| 12.5¢ | 106947 | 2 |
View Market Depth
| No. | Vol. | Price($) |
|---|---|---|
| 1 | 33402 | 0.115 |
| 2 | 9099 | 0.110 |
| 2 | 126900 | 0.105 |
| 5 | 543009 | 0.100 |
| 1 | 30303 | 0.099 |
| Price($) | Vol. | No. |
|---|---|---|
| 0.125 | 106947 | 2 |
| 0.130 | 323620 | 3 |
| 0.135 | 230549 | 8 |
| 0.140 | 155336 | 3 |
| 0.145 | 13227 | 4 |
| Last trade - 15.41pm 12/09/2025 (20 minute delay) ? |
Featured News
| SPL (ASX) Chart |